What Is Serialization in the Pharmaceutical Sector?
Life saver: drug serialization
The illegal trade in counterfeit products is a sad part of everyday life in the pharmaceutical business. With the help of a serialization solution, this problem can be eliminated in the long term. But what exactly is serialization in drug production? - Learn more about it in our blog article.

The illegal business with counterfeit medicines is a global problem. Not only manufacturers and markets are affected, but also each of us as a consumer. Drug serialization comes into play for adequate protection against counterfeiting. The packaging is serialized - data matrix codes, a Tamper Evident seal, and other solutions are used.
The serialization of prescription drugs falls into the regulatory area. With the EU Anti-Counterfeiting Directive of 2011 and the Delegated Regulation (EU) 2016/161, the legal framework was created to ensure the traceability of precisely these medicines. For manufacturers of pharmaceutical products, there is no way around serialization software and instead, in the best case, towards Arvato CSDB.
Serialization in the Eurpean Union
The Arvato Corporate Serialization Database (Arvato CSDB) connects all elements of serialization. Starting with the serial number - this is printed as a data matrix code on the packaging of the medication. The code also contains a product code, the expiration date, and the designation of the production batch. Based on the national pharmaceutical product number, the product code is generated into the NTIN (National Trade Item Number) or GTIN (Global Trade Item Number). The article number is decisive for the trade of pharmaceuticals. The numbers produced by the database are unique and ensure complete drug serialization.
With the help of Arvato CSDB, all steps of drug serialization can be linked, and interfaces to suppliers, production sites and lines, and verification systems can be created. Thanks to end-to-end verification systems used throughout the EU, flawless and safe dispensing of medicines can be guaranteed.
Authenticity Verification in the Pharmacy
Before pharmaceutical products enter the distribution chain, the serial number stored in the data matrix code must be transferred by the companies to the pharmaceutical industry database. If the product is sold in the pharmacy, it must first be scanned and checked for authenticity. During the scan, an anonymized query is sent to the verification system. If the query is positive, the serial number is available in the database and the drug may be dispensed. The status of the serial number then changes so that the product cannot be sold again. Due to the separation of the database systems, stakeholders involved cannot see which pharmacy dispensed which drug. The following graphic provides a good overview of the process:
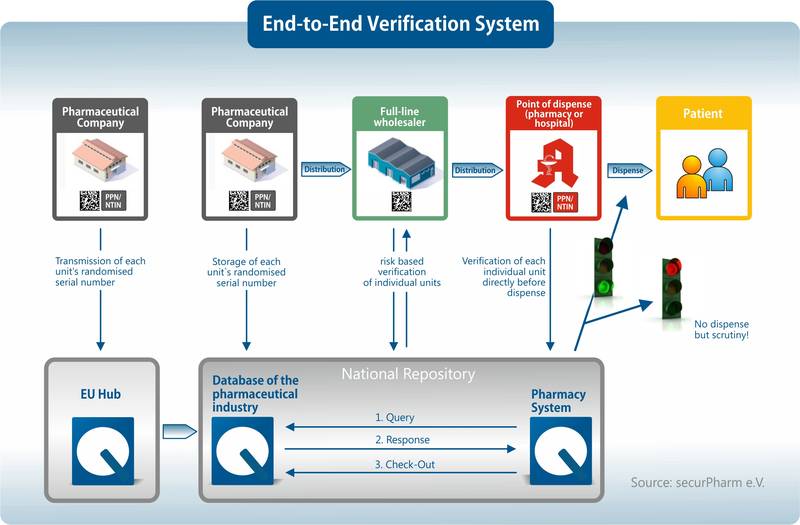
What happens if the scan at a pharmacy receives a negative response? - Pharmaceutical companies are obliged to check every report and make sure whether it is a counterfeit product. In most cases, the error comes from somewhere else. You can read more about this in our blog post Alert Management – Big Pending Task in Serialization.
Written by

Dr. Mareike Elgner holds a doctorate in biochemistry and is a key account manager at Arvato Systems. She focuses on digital solutions for the life science industry, particularly drug serialization. She also supports her customers in areas such as cloud and AI.